This article explores the concept of Quality by Design (QbD) in the pharmaceutical machinery industry. QbD is an approach that focuses on the systematic development and design of products and processes to ensure their quality. By implementing QbD principles, pharmaceutical machinery manufacturers can enhance the quality, reliability, and efficiency of their equipment, ultimately benefiting the entire pharmaceutical industry.
1) Implementing Quality by Design (QbD) Principles in Pharma Machinery
As a pharmaceutical industry professional, I believe it is crucial to implement Quality by Design (QbD) principles in the machinery utilized for pharmaceutical manufacturing processes. QbD is a systematic approach that ensures quality is built into every stage of the product development and manufacturing process. By incorporating QbD principles into the design and operation of machinery, we can ensure that the final product meets the required standards of safety, efficacy, and quality. This approach not only minimizes the risk of product failures and recalls but also enhances process efficiency and reduces manufacturing costs. Additionally, QbD principles encourage innovation and continuous improvement in the pharmaceutical industry, leading to the development of safer and more effective medicines for patients worldwide.
2) Benefits of Quality by Design (QbD) Approach in Pharma Machinery
As a pharmaceutical industry professional, I cannot stress enough the importance of the Quality by Design (QbD) approach in our machinery. The benefits it brings to the table are truly remarkable. Firstly, QbD helps us in identifying and understanding critical process parameters, allowing us to optimize our production processes. This results in improved efficiency, reduced waste, and cost savings. Additionally, this approach enables us to proactively identify and mitigate risks, ensuring the safety and efficacy of our products. QbD also promotes a culture of continuous improvement, as it provides a systematic framework for monitoring and optimizing our processes over time. Overall, embracing QbD in pharmaceutical machinery is crucial for ensuring high-quality, reliable, and cost-effective production.
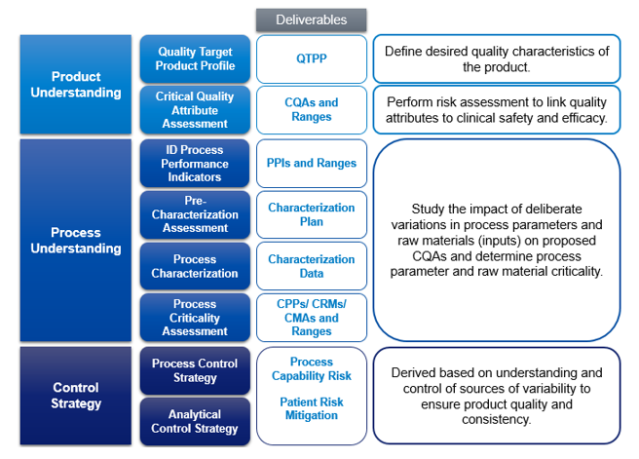
3) Key Components of Quality by Design (QbD) in Pharma Machinery
When it comes to ensuring the quality of pharmaceutical machinery, there are several key components of Quality by Design (QbD) that need to be considered. Firstly, it is essential to have a comprehensive understanding of the manufacturing process and the critical parameters that influence the quality of the final product. This includes factors such as the materials used, the equipment being used, and the specific steps involved in the production process. Additionally, it is crucial to establish a robust risk management system to identify and mitigate any potential risks that may arise during the manufacturing process. Finally, effective communication and collaboration between all stakeholders, including manufacturers, regulatory authorities, and healthcare professionals, are vital to ensure that the highest quality standards are met. By incorporating these key components of QbD, pharmaceutical companies can enhance the quality and reliability of their machinery, ultimately leading to safer and more effective medications for patients.
4) Challenges in Adopting Quality by Design (QbD) in Pharma Machinery
As a quality control professional in the pharmaceutical industry, I understand the challenges that arise when trying to adopt Quality by Design (QbD) in pharma machinery. QbD is a systematic approach that aims to ensure product quality by designing quality into the manufacturing process rather than relying solely on end-product testing. However, implementing QbD in pharma machinery can be complex. One of the main challenges is the lack of standardized guidelines and regulations specific to QbD implementation. This makes it difficult to establish a consistent framework across different manufacturers. Additionally, there may be resistance to change from employees who are accustomed to traditional quality control methods. Overcoming these challenges requires collaboration between industry experts, regulatory bodies, and manufacturers to develop comprehensive guidelines and provide the necessary training and education for all stakeholders. With the right support and commitment, the adoption of QbD in pharma machinery can greatly enhance product quality and ultimately benefit patients.
5) Case Studies: Successful Implementation of Quality by Design (QbD) in Pharma Machinery
In my experience as a Quality Assurance Manager in the pharmaceutical industry, I have witnessed the successful implementation of Quality by Design (QbD) in various machinery processes. One notable case study involved the production of tablets using a high-speed tablet press machine. By utilizing QbD principles, we were able to optimize and control critical parameters such as tablet weight, hardness, and dissolution rate. This allowed us to consistently manufacture high-quality tablets that met strict regulatory requirements. The use of QbD not only improved the overall efficiency of the process but also minimized the risk of batch failures and wastage. Through this case study, it became evident that implementing QbD in pharmaceutical machinery can lead to significant improvements in product quality and process reliability.
6) Future Trends in Quality by Design (QbD) for Pharma Machinery
In my opinion, the future trends in Quality by Design (QbD) for Pharma Machinery are extremely promising. As technology continues to advance at a rapid pace, we can expect to see more sophisticated and efficient machinery being developed for the pharmaceutical industry. This will not only improve the quality and safety of medical products but also streamline the manufacturing process, leading to shorter production times and reduced costs. Additionally, there is a growing emphasis on data-driven decision-making in the pharmaceutical sector, which will undoubtedly impact the design of future Pharma Machinery. With the integration of advanced analytics and artificial intelligence, manufacturers will have access to real-time insights and predictive modeling, allowing them to proactively detect and address potential quality issues. Overall, the future of QbD for Pharma Machinery looks incredibly promising and will greatly benefit both manufacturers and patients.
Conclusion
In conclusion, Quality by Design (QbD) in pharma machinery is a crucial aspect for ensuring the production of safe and effective pharmaceutical products. By integrating QbD principles into the design, development, and manufacturing processes of machinery, pharmaceutical companies can enhance product quality, reduce risks, and improve overall efficiency. Therefore, implementing QbD in pharma machinery is not only beneficial for the industry but also for the patients who rely on these medications for their health and well-being.
What is Quality by Design (QbD) in Pharma Machinery?
Quality by Design (QbD) is a systematic approach that emphasizes the importance of understanding pharmaceutical product and process performance variability. It involves identifying critical quality attributes (CQAs) and critical process parameters (CPPs) during the development of pharmaceutical machinery.
Why is Quality by Design (QbD) important in Pharma Machinery?
QbD helps in designing and developing robust pharmaceutical machinery that consistently meets the desired quality standards. It allows for early identification and control of potential sources of variability, thus reducing the risk of product failure and ensuring patient safety.
What are the benefits of implementing Quality by Design (QbD) in Pharma Machinery?
Implementing QbD principles in Pharma Machinery brings several benefits, including improved process understanding, increased process efficiency, enhanced product quality, reduced manufacturing costs, and improved regulatory compliance.
How is Quality by Design (QbD) implemented in Pharma Machinery?
QbD is implemented in Pharma Machinery through a series of steps, including identifying critical quality attributes (CQAs), selecting critical process parameters (CPPs), establishing the design space, utilizing risk-based approaches, performing process validation, and continuously monitoring and improving the process.
What are critical quality attributes (CQAs) and critical process parameters (CPPs)?
Critical quality attributes (CQAs) are the physical, chemical, biological, and microbiological properties or characteristics that determine the performance and quality of a pharmaceutical product. Critical process parameters (CPPs) are the key variables that can affect the CQAs and should be controlled within established limits to ensure the desired product quality.
How does Quality by Design (QbD) impact regulatory compliance in Pharma Machinery?
QbD provides a systematic and scientific approach to pharmaceutical development and manufacturing, which aligns with regulatory expectations. By implementing QbD, manufacturers can demonstrate a thorough understanding of their processes and a proactive approach to ensuring product quality, thus enhancing regulatory compliance.

